Vitamin B2 Metabolism Shields Cancer Cells from Ferroptosis, Study Finds
Researchers have uncovered a crucial mechanism that helps cancer cells survive, revealing that vitamin B2, also known as riboflavin, allows tumors to resist a form of cell death called ferroptosis. The discovery could open the door to new therapies for treatment-resistant cancers.
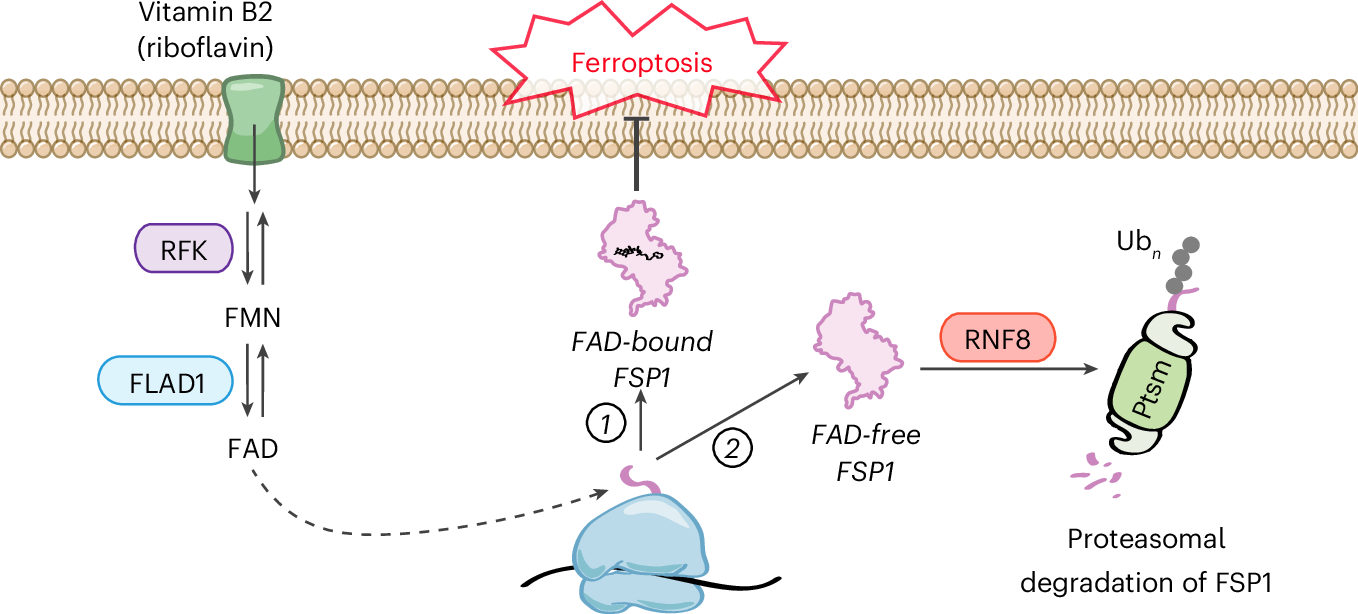
Ferroptosis is a unique type of cell death caused by the accumulation of toxic lipid molecules that damage cell membranes. Scientists have increasingly focused on ferroptosis as a potential way to kill stubborn cancer cells that evade traditional therapies. However, this new research demonstrates that cancer cells can manipulate vitamin B2 metabolism to protect themselves from this process.
According to the study, when cancer cells metabolize vitamin B2, they generate molecules that stabilize a key protein called ferroptosis suppressor protein 1 (FSP1). FSP1 neutralizes harmful lipid peroxides, preventing ferroptosis from occurring. By keeping this protein active, cancer cells effectively shield themselves from one of the body’s natural cell death mechanisms, allowing tumors to survive even under treatment or stress.
“This is a remarkable finding,” said Dr. Elena Novak, a cancer biologist not involved in the study. “Targeting the vitamin B2 metabolic pathway could make cancer cells more vulnerable to ferroptosis-based therapies, which may help overcome resistance in aggressive tumors.”
The discovery also underscores the complexity of cancer metabolism. While vitamin B2 is essential for the health of normal cells, cancer cells exploit it to their advantage, creating a challenge for researchers developing therapies that selectively target tumors without harming healthy tissue. Scientists are now exploring drugs that could interfere with riboflavin metabolism or inhibit FSP1 specifically in cancer cells, potentially opening a new front in cancer treatment.
Experts believe that understanding how tumors manipulate nutrients like vitamin B2 is critical to designing next-generation therapies. The findings could lead to treatments that combine metabolic disruption with existing chemotherapy or immunotherapy, increasing the chances of killing cancer cells that previously resisted treatment.
While clinical applications are still in development, this research provides a promising roadmap for targeting one of cancer’s survival strategies. By exploiting the dependency of tumors on vitamin B2 metabolism, scientists hope to make cancer cells more susceptible to ferroptosis and improve outcomes for patients with hard-to-treat cancers.